
The ocean’s average pH is now around 8.1 offsite link, which is basic (or alkaline), but as the ocean continues to absorb more CO 2, the pH decreases and the ocean becomes more acidic. Water and carbon dioxide combine to form carbonic acid (H 2CO 3), a weak acid that breaks (or “dissociates”) into hydrogen ions (H +) and bicarbonate ions (HCO 3 -).īecause of human-driven increased levels of carbon dioxide in the atmosphere, there is more CO 2 dissolving into the ocean. Carbon dioxide and seawaterĬarbon dioxide, which is naturally in the atmosphere, dissolves into seawater. The pH scale is an inverse of hydrogen ion concentration, so more hydrogen ions translates to higher acidity and a lower pH. Anything higher than 7 is basic (or alkaline) and anything lower than 7 is acidic. The pH scale runs from 0 to 14, with 7 being a neutral pH. This process has far reaching implications for the ocean and the creatures that live there. When CO 2 is absorbed by seawater, a series of chemical reactions occur resulting in the increased concentration of hydrogen ions. As levels of atmospheric CO 2 increase from human activity such as burning fossil fuels (e.g., car emissions) and changing land use (e.g., deforestation), the amount of carbon dioxide absorbed by the ocean also increases. The ocean absorbs about 30% of the carbon dioxide (CO 2) that is released in the atmosphere. 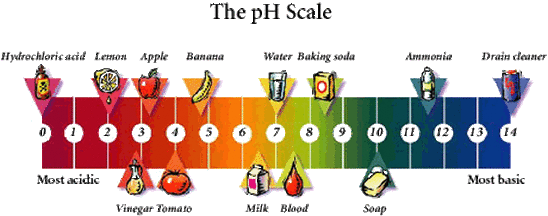 
About the education resource collections.NOAA Sea to Sky: Education resource database.NOAA in your backyard: Education contacts near you.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
